Amino Trimethylene Phosphonic Acid Solution
- Loading Port:
- Tianjin
- Payment Terms:
- TT OR LC
- Min Order Qty:
- -
- Supply Capability:
- 6000 m.t./month
OKorder Service Pledge
OKorder Financial Service
You Might Also Like
Product Description:
Amino tris(methylene phosphonic acid) / Amino Trimethylene Phosphonic Acid/ ATMP / 6419-19-8 / C3H12NO9P3
CAS No. 6419-19-8
Molecular Formula: N(CH2PO3H2)3
Molecular weight: 299.05
Structural Formula: 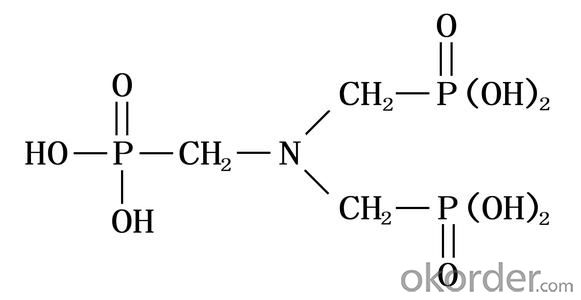
Properties:
ATMP has excellent chelation, low threshold inhibition and lattice distortion ability. It can prevent scale formation, calcium carbonate in particular, in water system. ATMP has good chemical stability and is hard to be hydrolyzed in water system. At high concentration, it has good corrosion inhibition.
ATMP is used in industrial circulating cool water system and oilfield water pipeline in fields of thermal power plant and oil refinery plant. ATMP can decrease scale formation and inhibit corrosion of metal equipment and pipeline. ATMP can be used as chelating agent in woven and dyeing industries and as metal surface treatment agent.
The solid state of ATMP is crystal powder, soluble in water, easily deliquescence, suitable for usage in winter and freezing districts. Because of its high purity, it can be used in woven & dyeing industries and as metal surface treatment agent.
Specification:
| Items | Index | |
|---|---|---|
| Standard | Solid | |
| Appearance | Clear, Colorless to pale yellow aqueous solution | White crystal powder |
| Active acid % | 50.0-51.0 | 95.0min |
| Chloride (as Cl-)% | 1.0 max | 1.0 max |
| pH value (1% solution) | 2.0 max | 2.0 max |
| Fe,mg/L | 10.0max | 20.0max |
| Density (20°C)g/cm3 | 1.31-1.35 | - |
| Colour APHA (Hazen) | 30.0max | - |
Application range&using method:
ATMP is usually used together with other organophosphoric acid, polycarboxylic acid and salt to built all organic alkaline water treatment agent. ATMP can be used in many different circulating cool water system. The recommended dosage is 5-20mg/L. As corrosion inhibitor, The recommended dosage is 20-80mg/L.
Package and Storage:
ATMP liquid: Normally In 30kg or 250kg net Plastic Drum;ATMP solid: 25kg inner liner polyethylene (PE) bag, outer plastic woven bag, or confirmed by clients request.Storage for ten months in room shady and dry place.
Safety Protection:
ATMP is Acidity, Avoid contact with eye and skin, once contacted, flush with water.
Shipping Date: Within 7-10 workdays after receiving your deposit.
Our Service:
Own Lab and joint venture factory.
Superb r&d team;Safety standardization production.
Rich experience in export and strong logistical support.
Good relationship with many large domestic pharmaceutical factory.
Perfect service, perfect supply chain.
- Q: Have you Read it ? If SoCan You Think Of Any Good Group Discussion Questions ?
- Discussion and Essay Questions What is a catalyst? Who are the catalysts in this book? Why? What is Kate running from? How did she get lost (literally and figuratively)? Why does Kate hate English class? What are some of the myths surrounding the college application process? About high school? About kids like Kate? Kids like Teri? Find the list of vocabulary words given out for homework in Kate's English class. The author deliberately chose them. What is their significance? Why do authors do things like that? Does this make you hate authors? (joke... just wanted to see if you're paying attention) Discuss these image systems (symbols) found in the book: a. chemistry b. periodic table c. vision/sight d. movement vs. being stuck e. electricity What examples of safety awareness and danger can you find? How are they connected to each other? Make a list of all the elements and chemical terms used as chapter headings in the book. Explain how the chapters headings refer to the action with each chapter. Find the hints of Alice in Wonderland in the book. How does the story of Alice's journey reflect Kate's? Catalyst takes place in Merryweather High School, also the setting for Speak. What connections link the two books? How is food used in this book? Why don't we learn more about the life of Teri Litch (hint: examine the book's POV) How is Kate's relationship with her father? Why is it that way? Does it change over the course of the book. (Give examples for each answer.) Discuss the role of religion in this book. What does Kate believe in? Teri? Mitchell? How and why does Kate's relationship with Teri change after Mikey's death? Why are the three sections of the book labeled Solid, Liquid and Gas? Who is the most important character in this book? Why? How do the characters in Catalyst deal with defeat?
- Q: The catalyst before and after the reaction of the quality and nature of the same, does it mean that a little catalyst can be all the substrate reaction? Such as: one gram of MnO2 can reflect the infinite H2O2?
- There are positive catalyst and anti-catalyst, as well as the amount of catalyst is also related to the rate, not the more the better
- Q: The role of catalyst in chemical reactions
- Negative catalysts can be used to control the reaction rate (such as some reaction too fast, instantly release a lot of energy caused by danger, you can join), common is the antioxidant
- Q: The size of △ H in the thermochemical reaction equation is related to the use and unused catalyst
- It does not matter
- Q: Will the catalyst change in the chemical reaction?
- In fact, there are two kinds of catalyst mechanism, one is not a chemical reaction, the reaction did not occur throughout the chemical reaction, the catalyst did not change; one is involved in the reaction process, but the final production of the catalyst, which The class reaction generally occurs in a stepwise chemical reaction.
- Q: What is a chemical catalyst?
- A substance that participates in chemical reactions but does not change the quality and chemical properties. It can change the reactant activation energy, speed up or slow down the reaction rate.
- Q: What makes an enzyme a catalyst?
- D: it quickens a chemical reaction yet isn't completely replaced by skill of the reaction this is a standard definition of the understanding catalyst that a biologist gave: A chemical/substance that alters the fee of a chemical reaction yet isn't used up interior the technique.
- Q: I need to name 3everyday catalysts for my chemistry homework. I don't want anything complicated there must just be 3 easy ones out there... Then could you please state what each one is used for? Please do it like this, e.g.Hello ; used to say hi to someone. I know that's a bad example but please!!
- Hello ; Chemical catalysts are substances that increase the rate of reaction (while remaining themselves chemically unchanged: Polyethylene, the polymer used to make everything from: 1. garbage bags and 2. grocery bags, to 3. squeezable bottles, to 4. cable insulation, is made by passing ethylene gas over a catalyst. Most other polymers are made in the same (or similar) way. Synthetic rubber, nylon, polyester, PVC, teflon, etc. It's not hard to think of 10 uses for these catalyst-produced materials alone... e.g. : 5. rain coats 6. pantyhose 7. water pipes 8. bouncy balls 9. non-stick surfaces and pads 10. saran wrap 11. food containers 12. cell-phone and laptop cases 13. cheap wigs 14. fishing line... 15. Enzymes are Biological catalysts: 16. beer and 17. bread are typically made with yeast, a living organism containing enzymes I hope this helps!
- Q: how a catalyst can provide a new route in forming the product?
- A catalyst will almost always provide an alternative route for the reaction in question. It is common to hear the statement that a catalyst does not take part in a reaction, but that is almost always untrue. Properly defined, a catalyst is a species that alters the rate of a chemical reaction and which can be recovered unchanged BY MASS at the conclusion. In other words, the amount of catalyst present at the end of the reaction is the same as the amount at the start, but the catalyst itself has likely reacted many many times as the reaction proceeded. A simple example is the decomposition of hydrogen peroxide. The reaction taking place can be summarised as: 2 H2O2(l) ---2 H2O(l) + O2(g) This reaction is very slow under normal circumstances. However, if you drop in a crystal of potassium iodide, the reaction becomes extremely fast. The reason is that the potassium iodide dissolves and dissociates releasing iodide ions. These provide a new two-step reaction pathway in which both steps are rapid: H2O2 + I- ---H2O + IO- H2O2 + IO- ---H2O + O2 + I- Notice that if you add these equations together you get the same overall reaction as before. The iodide ion does not take part in the overall reaction, and so must be present at the end. However, it does take part in the mechanism of the reaction, and is undergoing a lot of reactions.
- Q: Can you describe at least 4 ways a catalyst can lower the activation energy of a reaction?
- To see how a catalyst accelerates the reaction, we need to look at the potential energy diagram shown below which compares the non-catalytic and the catalytic reaction. For the non-catalytic reaction, the figure is simply the familiar way to visualize the Arrhenius equation: the reaction proceeds when A and B collide with succificient energy to overcome the activation barrier. The change in Gibbs free energy between reactants, A + B, and the product P is delta G. The catalytic reaction starts by bonding of the reactants A and B to the catalyst, in a spontaneous reaction. Hence, the formation of this complex is exothermic and the free energy is lowered. There then follows the reaction between A and B while they are bound to the catalyst. This step is associated with an activation energy; however, it is significantly lower than that for the uncatalyzed reaction. Finally, the product P seperates from the catalyst in an endothermic step. The energy diagram illustrates 4 ways the catalyst works : The catalyst offers an alternative path for the reaction that is energetically more favorable The activation energy of the catalytic reaction is significantly smaller than that of the uncatalyzed reaction; hence the rate of the catalytic reaction is much larger The overall change in free energy for the catalytic reaction equals that of the uncatalyzed reaction. Hence, the catalyst does not affect the equilibrium constant for the overall reaction. A catalyst cannot change the thermodynamics of a reaction but it can change the kinetics. The catalyst accelerates both the forward and the reverse reaction to the same extent. In other words, if a catalyst accelerates the formation of product P from A and B, it will do the same for the decomposition of P into A and B.
Send your message to us
Amino Trimethylene Phosphonic Acid Solution
- Loading Port:
- Tianjin
- Payment Terms:
- TT OR LC
- Min Order Qty:
- -
- Supply Capability:
- 6000 m.t./month
OKorder Service Pledge
OKorder Financial Service
Similar products
Hot products
Hot Searches



















