Industrial Solid Amino Trimethylene Phosphonic Acid
- Loading Port:
- Tianjin
- Payment Terms:
- TT OR LC
- Min Order Qty:
- -
- Supply Capability:
- 6000 m.t./month
OKorder Service Pledge
OKorder Financial Service
You Might Also Like
Product Description:
Amino tris(methylene phosphonic acid) / Amino Trimethylene Phosphonic Acid/ ATMP / 6419-19-8 / C3H12NO9P3
CAS No. 6419-19-8
Molecular Formula: N(CH2PO3H2)3
Molecular weight: 299.05
Structural Formula: 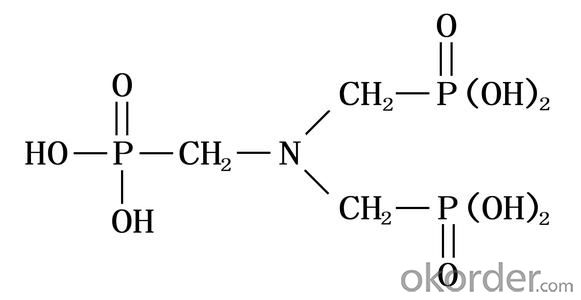
Properties:
ATMP has excellent chelation, low threshold inhibition and lattice distortion ability. It can prevent scale formation, calcium carbonate in particular, in water system. ATMP has good chemical stability and is hard to be hydrolyzed in water system. At high concentration, it has good corrosion inhibition.
ATMP is used in industrial circulating cool water system and oilfield water pipeline in fields of thermal power plant and oil refinery plant. ATMP can decrease scale formation and inhibit corrosion of metal equipment and pipeline. ATMP can be used as chelating agent in woven and dyeing industries and as metal surface treatment agent.
The solid state of ATMP is crystal powder, soluble in water, easily deliquescence, suitable for usage in winter and freezing districts. Because of its high purity, it can be used in woven & dyeing industries and as metal surface treatment agent.
Specification:
| Items | Index | |
|---|---|---|
| Standard | Solid | |
| Appearance | Clear, Colorless to pale yellow aqueous solution | White crystal powder |
| Active acid % | 50.0-51.0 | 95.0min |
| Chloride (as Cl-)% | 1.0 max | 1.0 max |
| pH value (1% solution) | 2.0 max | 2.0 max |
| Fe,mg/L | 10.0max | 20.0max |
| Density (20°C)g/cm3 | 1.31-1.35 | - |
| Colour APHA (Hazen) | 30.0max | - |
Application range&using method:
ATMP is usually used together with other organophosphoric acid, polycarboxylic acid and salt to built all organic alkaline water treatment agent. ATMP can be used in many different circulating cool water system. The recommended dosage is 5-20mg/L. As corrosion inhibitor, The recommended dosage is 20-80mg/L.
Package and Storage:
ATMP liquid: Normally In 30kg or 250kg net Plastic Drum;ATMP solid: 25kg inner liner polyethylene (PE) bag, outer plastic woven bag, or confirmed by clients request.Storage for ten months in room shady and dry place.
Safety Protection:
ATMP is Acidity, Avoid contact with eye and skin, once contacted, flush with water.
Shipping Date: Within 7-10 workdays after receiving your deposit.
Our Service:
Own Lab and joint venture factory.
Superb r&d team;Safety standardization production.
Rich experience in export and strong logistical support.
Good relationship with many large domestic pharmaceutical factory.
Perfect service, perfect supply chain.
- Q: Before and after the reaction, the chemical properties and quality of the water did not change, and the water was the catalyst
- 3I2 + 3H2O = 3HI + 3HIO
- Q: Chemical catalyst system baa?
- Can speed up or slow down the reaction rate without participating in the reaction of the material
- Q: The addition of the catalyst has no effect on the chemical equilibrium of the movement
- Hello, the essence of chemical equilibrium is a dynamic equilibrium, under certain conditions, the equilibrium constant of the reaction is a certain value, the role of the catalyst is to reduce the activation energy required for the reaction, increase the number of activated molecules, so that the reaction within a unit time The number of molecules increased, but the positive reaction is positive for the reaction, so do not change the speed
- Q: Hydrogen and oxygen in the role of the catalyst can do the chemical formula of aviation fuel?
- 2H2 + O2 = catalyst = 2H2O
- Q: What is the similarity between enzymes and general chemical catalysts?
- (1) The enzyme is the same in many respects as a biocatalyst and a general catalyst, such as a small amount and a high catalytic efficiency. As with the general catalyst, the enzyme can only change the rate of chemical reaction and does not change the equilibrium of the chemical reaction It is possible to catalyze the activation of a large number of substrates in a short time and to reflect the high efficiency of enzyme catalysis.The enzyme can reduce the activation energy of the reaction (activation) (△ G) during the reaction, but the reaction rate is accelerated and the reaction time is reduced, but the equilibrium constant is not changed. (2) However, the enzyme is a biological macromolecule (1) Enzyme-catalyzed high efficiency: The catalytic effect of the catalyst can increase the reaction rate by 10 ^ 6 ~ 10 ^ 12 times, which is at least several times higher than that of the conventional catalyst. (2) The enzyme catalyst Highly specificity: including specificity of response, substrate specificity, chirality specificity, geometric specificity, etc., that an enzyme can only act on a certain class or a specific substance. Bond, ester bond, peptide bond and so on can be catalyzed by acid-base hydrolysis, but the hydrolysis of these chemical bonds are different, respectively, the corresponding glycosidase, esterase and peptidase, that is, they were specific (3) enzymatic reaction conditions are mild: enzymatic reaction is generally carried out in aqueous solution of pH = 5 ~ 8, the reaction temperature range is 20 ~ 40 ℃
- Q: Is the catalyst in the chemical reaction better?
- No, you can. Too much useless
- Q: I opened catalyst control center to see what its was, i didn't change anything and now when i full screen a youtube and hulu the screen will be black and the loading bar at the bottom will be stuck. I also noticed that now when i play minecraft all the blocks have a black out line i cant get rid of. I tried deleting my .minecraft and that didn't fix it.
- on the backside suitable hand corner of the small reveal, there's a field with a smaller field in it (suitable of the quantity), press that, it provides you with the finished reveal. you could press the comparable button once you opt for to bypass decrease back. The button would have a crimson X in it this time.
- Q: explain how a catalyst can affect the rate of reaction but not be in the overall equation.?
- Simply, the catalyst provides a better way for the reaction to occur (some won't work without the catalyst) and usually a favorable reaction will run faster if there is a catalyst. There are many ways for this to happen. The catalyst can provide more surface area for a reaction to occur, it can do an adsorption process where one of the reactants sticks to the surface and exposes a portion of the molecule which is more favorable to the reaction. It can lower the energy required for the reaction to occur (same effect as increasing temperature) by favoring an intermediate step in the reaction. Lots of different ways, some not fuly identified or understood. The catalyst people who work with the platinum metals groups are notorious for keeping their mixtures secret. A better gasoline catalyst for refineries is worth billions in profits. It is not considered in the equation because you get back what you start with even if one of the intermediate steps involves changing the chemical composition of the catalyst and then has it returning to its original state with the formation of the product.
- Q: Does anybody have any tips or references I can go to for this? I'm writing a novel and the main character wants to be a catalyst; the story isn't about him and his journey, so much as the effects on everyone around him that come about simply because of him being there and being who he is. How would I go about doing this, and doing it well?
- Ways to be a catalyst: By his behaviour: - He is a good listener. People use him as a sounding board and make important decisions as a result. - He is indiscreet and inadvertently reveals people's secrets and back-sniping comments. - He is a ****-stirring gossip. - He is a home-wrecking Lothario. - He is wise and gives good advice. - He is a hooligan who ruins businesses with vandalism, costs people their jobs and ruins relationships by beating people up. - He is a manipulative, blackmailing bastard who tries to turn every woman into a prostitute and con every man out of his money. - He is a charlatan who preaches nonsense about religion, health and business investments. By effortlessly influencing other people's behaviour: - He is famous and people try to impress him wherever he goes. - He has cancer or a disabling war wound. People admire and pity him and are shocked by his PTs mood wings. - He is gay, Muslim or a suspected paedophile and people want to persecute or cure him. - He is destitute. People argue amongst themselves over whether it's because he's lazy, has bad karma or there but for the grace of God go I. - He really looks like Jesus, Buddha or Santa and the sight of him makes people contemplate their Humanist values, coming to various conclusions. Perhaps you can write about his reputation; how he earned it, how it precedes him and provokes prejudices that he sometimes confirms or disproves. Use a detached, omniscient God-narrator. (Pretend Morgan Freeman's reading the audiobook.) Alternatively perhaps he is not a catalyst but a neurotic voyeur, fascinated by the minutiae of other people's lives so that the mundane appears tumultuous. He is not influencing people but your account of the changes in people's lives are centred on his observation of them.
- Q: An important property of the catalyst is that the reaction equilibrium is not changed while increasing the forward reaction rate and the reverse reaction rate. However, because the enzyme for the specificity of the substrate, is not almost every reaction by the enzyme are one way to do it.
- In biochemical reactions, most appear to be positive / inverse reaction combinations, in fact there are differences in peripheral product / energy offerings. In addition to the molecules of interest, there are many other small molecules involved in the reaction, and these small molecules in the forward or reverse reaction in the transformation is not completely mutually negative. Even for the same reaction, since the corresponding substrate may be removed in the subsequent step and the pulling balance occurs, it is also possible that the forward or reverse direction can occur. Decisive factors usually come from the surrounding other enzymes, coenzymes, small molecules concentration.
Send your message to us
Industrial Solid Amino Trimethylene Phosphonic Acid
- Loading Port:
- Tianjin
- Payment Terms:
- TT OR LC
- Min Order Qty:
- -
- Supply Capability:
- 6000 m.t./month
OKorder Service Pledge
OKorder Financial Service
Similar products
Hot products
Hot Searches






















