Amino Trimethylene Phosphonic Acid National Stardard Quality
- Loading Port:
- Tianjin
- Payment Terms:
- TT OR LC
- Min Order Qty:
- -
- Supply Capability:
- 6000 m.t./month
OKorder Service Pledge
OKorder Financial Service
You Might Also Like
Product Description:
Amino tris(methylene phosphonic acid) / Amino Trimethylene Phosphonic Acid/ ATMP / 6419-19-8 / C3H12NO9P3
CAS No. 6419-19-8
Molecular Formula: N(CH2PO3H2)3
Molecular weight: 299.05
Structural Formula: 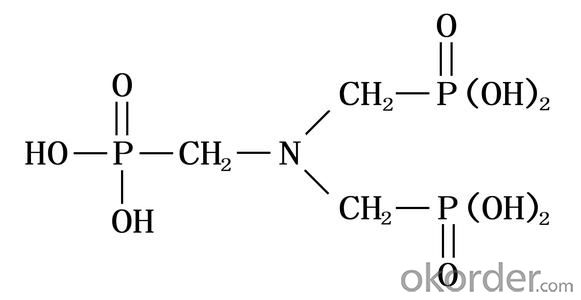
Properties:
ATMP has excellent chelation, low threshold inhibition and lattice distortion ability. It can prevent scale formation, calcium carbonate in particular, in water system. ATMP has good chemical stability and is hard to be hydrolyzed in water system. At high concentration, it has good corrosion inhibition.
ATMP is used in industrial circulating cool water system and oilfield water pipeline in fields of thermal power plant and oil refinery plant. ATMP can decrease scale formation and inhibit corrosion of metal equipment and pipeline. ATMP can be used as chelating agent in woven and dyeing industries and as metal surface treatment agent.
The solid state of ATMP is crystal powder, soluble in water, easily deliquescence, suitable for usage in winter and freezing districts. Because of its high purity, it can be used in woven & dyeing industries and as metal surface treatment agent.
Specification:
| Items | Index | |
|---|---|---|
| Standard | Solid | |
| Appearance | Clear, Colorless to pale yellow aqueous solution | White crystal powder |
| Active acid % | 50.0-51.0 | 95.0min |
| Chloride (as Cl-)% | 1.0 max | 1.0 max |
| pH value (1% solution) | 2.0 max | 2.0 max |
| Fe,mg/L | 10.0max | 20.0max |
| Density (20°C)g/cm3 | 1.31-1.35 | - |
| Colour APHA (Hazen) | 30.0max | - |
Application range&using method:
ATMP is usually used together with other organophosphoric acid, polycarboxylic acid and salt to built all organic alkaline water treatment agent. ATMP can be used in many different circulating cool water system. The recommended dosage is 5-20mg/L. As corrosion inhibitor, The recommended dosage is 20-80mg/L.
Package and Storage:
ATMP liquid: Normally In 30kg or 250kg net Plastic Drum;ATMP solid: 25kg inner liner polyethylene (PE) bag, outer plastic woven bag, or confirmed by clients request.Storage for ten months in room shady and dry place.
Safety Protection:
ATMP is Acidity, Avoid contact with eye and skin, once contacted, flush with water.
Shipping Date: Within 7-10 workdays after receiving your deposit.
Our Service:
Own Lab and joint venture factory.
Superb r&d team;Safety standardization production.
Rich experience in export and strong logistical support.
Good relationship with many large domestic pharmaceutical factory.
Perfect service, perfect supply chain.
- Q: How are the 4 characteristics of a catalyst (1. organic or inorganic 2. reusable 3. Highly specific, and 4. lowers activation energy) important in preforming life functions? please be as specific as possible, i understand that these are characteristic, i just don't understand why they're beneficial, other than the reusable and lowers activation energy one.
- Organic or Inorganic - the catalyst (enzyme) must be organic to be found in the cell. Catalysts speed up chemical reactions inside a cell and must therefore be organic to be a functioning part of the cell. Reusable - There are so many reactions that catalysts are involved in that it would be a waste for the cell if a catalyst could only last one reaction, especially if there are inhibitors and competition for the active site. Catalysts must be reusable in order to keep the cell functioning. Catalysts always remain unchanged after a reaction. HIihly Specific - Catalysts are only made to catalyze one specific chemical reaction. Their active site has proteins bonded in such a way that only certain elements can enter the active site and H bond with those proteins. The fact that they are highly specific maximizes the productiveness of the cell. And it ensures that the cell only has catalysts to reactions that it needs to be completed. It also ensures that the elements are correctly bonded with eachother. If any two elements could enter the active site, there is no guarantee that the correct product will be produced. Catalysts and Enzymes must be super highly specific in order to properly function. Lowers Activation Energy - The more energy a cell has to spend to catalye a reaction, the worse it is for the cell and the less ATP is has for other reactions. Catalyts hold the substrates together so there is less energy that is needed to have the two substrates react with eachother. Activation Energy is the energy that is needed to start a reaction. So the less energy used by the cell for reactions, the better for the cell. Hope this helps
- Q: How does the catalyst affect chemical balance? Why the catalyst has no effect on the chemical equilibrium, for v-t diagram
- Chemical reaction to reactor molecules to reach a certain amount of energy in order to react, this energy is the activation energy of the reaction. While the material energy in nature can be obtained by probability statistics
- Q: Chemical equation if there is a catalyst and heating, which write in the equal sign above, which written in the following? Tomorrow academic level test, solution
- At the same time, the catalyst is written on, and the heating symbol is written under the equal sign. Only one is written on the equal sign
- Q: What is a Catalyst?
- Catalyst is a chemical substance which is used in chemical reactions in relatively small amounts to start up or increase the rate of the reaction without being consumed in the process. For example, Sulfuric Acid is used to dehydrate Ethanol to Ethylene. Enzymes in living beings are biological catalysts. Manganese dioxide, used to decompose Hydrogen Peroxide to Oxygen and Water. For detailed answer.... en.wikipedia.org/wiki/Catalysis
- Q: What is the PTC catalyst in chemistry?
- 1, polyether chain polyethylene glycol: H (OCH2CH2) nOH chain polyethylene glycol dialkyl ether: R (OCH2CH2) nOR2, cyclic crown ethers: 18 crown 6,15 crown 5, Fine and so on. 3, quaternary ammonium salt: commonly used quaternary ammonium salt phase transfer catalyst is benzyl triethyl ammonium chloride (TEBA), tetrabutyl ammonium bromide, tetrabutylammonium chloride, tetrabutylammonium hydrogen sulfate (TBAB) , Trioctylmethylammonium chloride, dodecyltrimethylammonium chloride, tetradecyltrimethylammonium chloride, and the like. 4, tertiary amine: R4N X, pyridine, tributylamine and the like. 5, quaternary ammonium base (its alkaline and sodium hydroxide similar) soluble in water, strong hygroscopicity. 6, quaternary phosphonium
- Q: What is the reaction in chemistry?
- Industrial production of ammonia, ethanol, industrial synthesis of ammonia, ethanol catalytic oxidation, acetaldehyde oxidation into acetic acid, ethyl acetate preparation, the transformation of automobile exhaust, benzene substitution reaction and addition reaction, some other addition reaction of hydrocarbons, Ethanol dehydration to produce ethylene and so on
- Q: What is the difference between a catalyst and an inducer in a chemical reaction?
- The catalyst does not participate in the reaction, but only the carrier of the reaction; the inducer will participate in the reaction
- Q: and can you give me an example of it .. please give it in easy terms if you can. thanks
- A okorder /... hope this helps!!
- Q: What is the catalyst called?
- Catalysts induce chemical reactions to change, cause chemical reactions to become faster or slower, or to react chemically at lower temperatures. The catalyst is also known as a catalyst in industry.
- Q: What kind of chemical reaction requires a catalyst?
- For example, the system of ammonia, S02 oxidation into SO3
Send your message to us
Amino Trimethylene Phosphonic Acid National Stardard Quality
- Loading Port:
- Tianjin
- Payment Terms:
- TT OR LC
- Min Order Qty:
- -
- Supply Capability:
- 6000 m.t./month
OKorder Service Pledge
OKorder Financial Service
Similar products
Hot products
Hot Searches



















