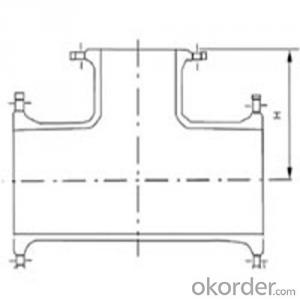
Coupling with Epoxy Coating
- Loading Port:
- China Main Port
- Payment Terms:
- TT OR LC
- Min Order Qty:
- -
- Supply Capability:
- -
OKorder Service Pledge
Quality Product, Order Online Tracking, Timely Delivery
OKorder Financial Service
Credit Rating, Credit Services, Credit Purchasing
You Might Also Like
1. Standards : ISO2531, ISO4422, EN545 , BS4772 , AWWA C110
2. material : GGG500-7 or other Ductile iron .
3. coating : Fusion bonded epoxy coating ; cement lining inside and zinc primer and bitumen painting outside ; bitumen painting inside and outside ; red anti-rust coating .
4. package : wooden cases with plastic layer , wooden pallets with plastic layer , steel crates .
5. accessories such as gaskets , bolts and nuts available upon request .
1. Standards : ISO2531, ISO4422, EN545 , EN598 , BS4772 , AWWA C110
2. material : GGG500-7 or other Ductile iron .
3. coating : Fusion bonded epoxy coating ;
4. package : wooden cases with plastic layer , wooden pallets with plastic layer , steel crates .
5. accessories such as gaskets , bolts and nuts available upon request .
2. material : GGG500-7 or other Ductile iron .
3. coating : Fusion bonded epoxy coating ; cement lining inside and zinc primer and bitumen painting outside ; bitumen painting inside and outside ; red anti-rust coating .
4. package : wooden cases with plastic layer , wooden pallets with plastic layer , steel crates .
5. accessories such as gaskets , bolts and nuts available upon request .
1. Standards : ISO2531, ISO4422, EN545 , EN598 , BS4772 , AWWA C110
2. material : GGG500-7 or other Ductile iron .
3. coating : Fusion bonded epoxy coating ;
4. package : wooden cases with plastic layer , wooden pallets with plastic layer , steel crates .
5. accessories such as gaskets , bolts and nuts available upon request .
- Q: sometimes i feel tired sometimes very tired. I went to bed about 11 and got up about qaurter to 7 so should i be tired or have i had lack of sleep? i am a vegetarian so i dont think i have enough iron is irin in fish? please help coz i hate feeling tired! lol
- For a vegetarian a good source of iron is nuts and some extra vitamin C will help to absorb more. Bread and cereals contain a bit of iron as well. There is very little iron in fish. If you are deficient in iron don't donate blood. If you are overloaded with iron, which can form iron plaque on your artery walls similar to cholesterol deposits, then do donate blood. Organic iron such as ferrous gluconate is absorbed better than inorganic iron such as ferrous sulphate, if you wish to take iron supplements.
- Q: what was significant about the discovery of iron oxide bands in the sedimentary layers?
- The conventional concept is that the banded iron layers were formed in sea water as the result of oxygen released by photosynthetic cyanobacteria, combining with dissolved iron in Earth's oceans to form insoluble iron oxides, which precipitated out, forming a thin layer on the substrate, which may have been anoxic mud (forming shale and chert). Each band is similar to a varve. The banding is assumed to result from cyclic variations in available oxygen. It is unclear whether these banded ironstone formations were seasonal or followed some other cycle. It is assumed that initially the Earth started out with vast amounts of iron dissolved in the world's acidic seas. Eventually, as photosynthetic organisms generated oxygen, the available iron in the Earth's oceans was precipitated out as iron oxides. At the tipping point where the oceans became permanently oxygenated, small variations in oxygen production produced pulses of free oxygen in the surface waters, alternating with pulses of iron oxide deposition
- Q: I have thick curly biracial hair and i need aperfect good all-round flat iron. is the RUSK one good what about the HAI? plz help. THX!
- It's alright, but the CHI is still the best.
- Q: I was wondering how good the straightening irons work
- Ehhh. I just got done with a Bedhead, and it was not a very good iron. I ended up buying a Chi. It's expensive, but definitely worth the price. The Bedhead irons are just a waste of money.
- Q: Even having something light to eat with it doesn‘t help much; only a full meal will prevent it.But why does iron irritate the stomach?
- Nothing to worry about. Copper gets like that from any exposure to the elements. As long as no water is leaking, I wouldn't worry about it. LOL Just look at the Statue of Liberty!!! I can't even imagine that she was once copper-colored.
- Q: i have iron deficiency ?
- No don't count on eating cornflakes. you need to take a dietary supplement called dessicated liver if you don't want to eat spinach, broccoli, lentils or beef.
- Q: I bought some ties from a department store and they have creases from how they were folded.Can you get the creases out with an iron?
- You definitely are a heterosexual. Meaning, you care a lot about your looks. Women that obsess about it, tend to be very superficial. It's not necessary to go through all the stuff you're doing. I am a woman, and trust me. Instead of spending 1000 hours a day on all that extra stuff, start spending time at the gym. If you get buff, women will DEFINITELY appreciate it. Not many women are attracted to very skinny men, simply because naturally we want to be protected (I'm not sure if you've taken biology). Spend time at the gym, of course look nice Showering 2x a day isn't necessary, unless you work out. Dressing up ALL the time, is a little weird. It shows you care a lot about looks, you should just wear normal clothes at least half the week. Otherwise, at least to me. You'll come off uptight. It's alright if your shoes are a little dirty ;) especially if you got the muscles.
- Q: Iron gate is the deepest gorge in Erope. But i want to know where is that lcated in Euroep.?
- Iron gate located in Romania the last gorge system on the Danube River, dividinate in g the Carpathian and Balkan mountains and forming part of the boundary between Serbia and Romania Coordinate 44° 40′ 16″ N, 22° 31′ 47″ E
Send your message to us
Coupling with Epoxy Coating
- Loading Port:
- China Main Port
- Payment Terms:
- TT OR LC
- Min Order Qty:
- -
- Supply Capability:
- -
OKorder Service Pledge
Quality Product, Order Online Tracking, Timely Delivery
OKorder Financial Service
Credit Rating, Credit Services, Credit Purchasing
Similar products
Hot products
Hot Searches
Related keywords